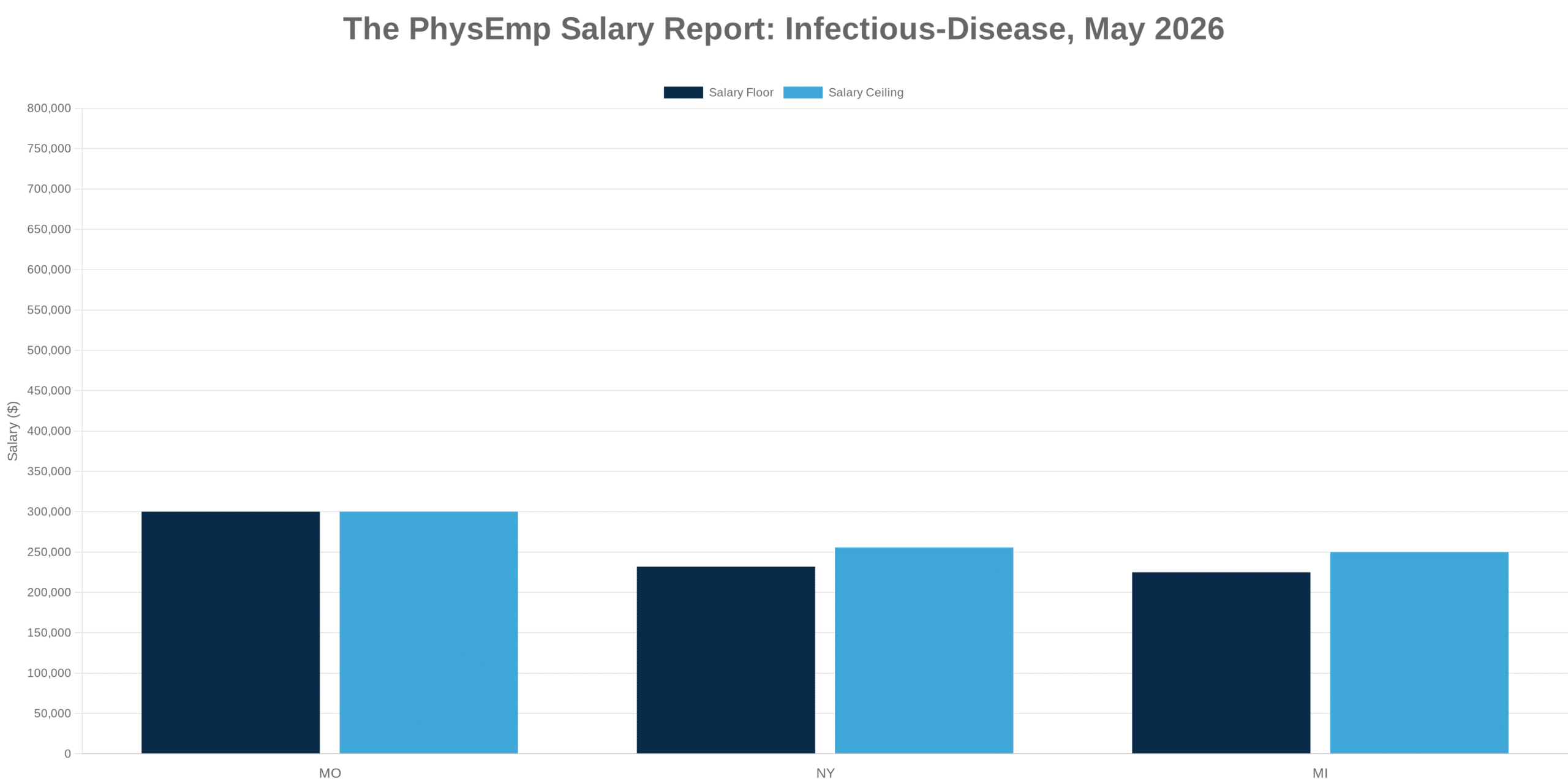
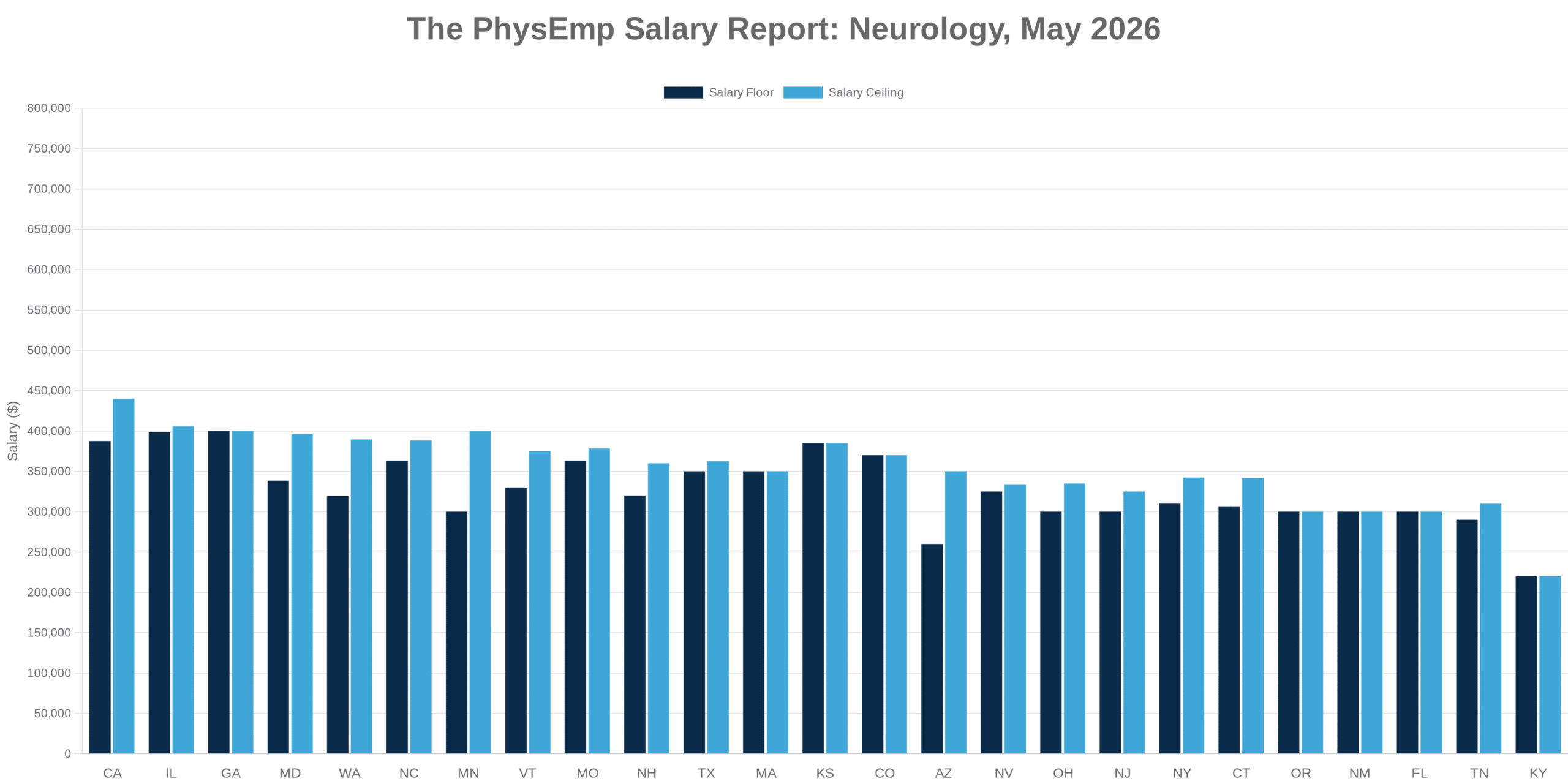
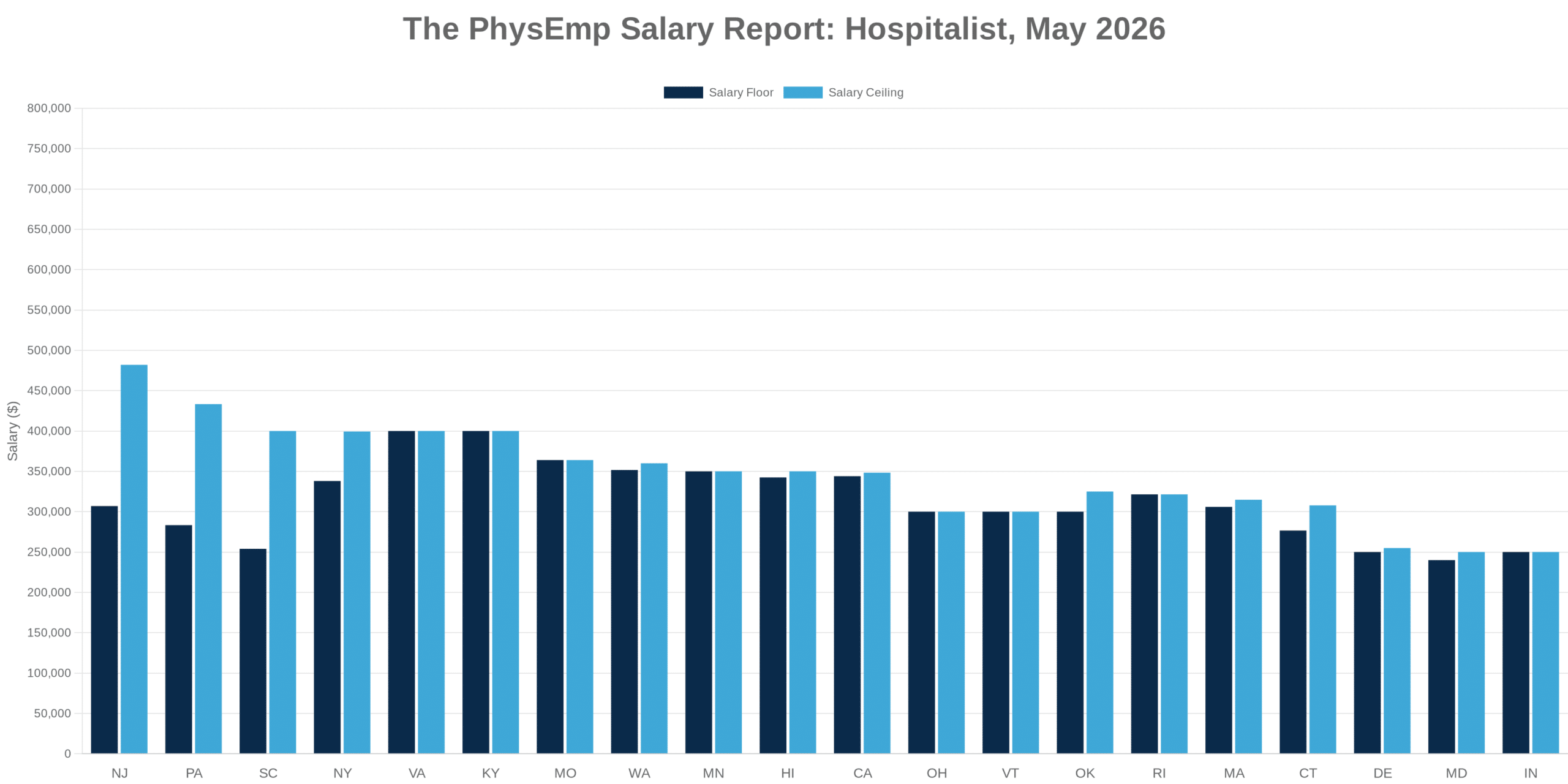
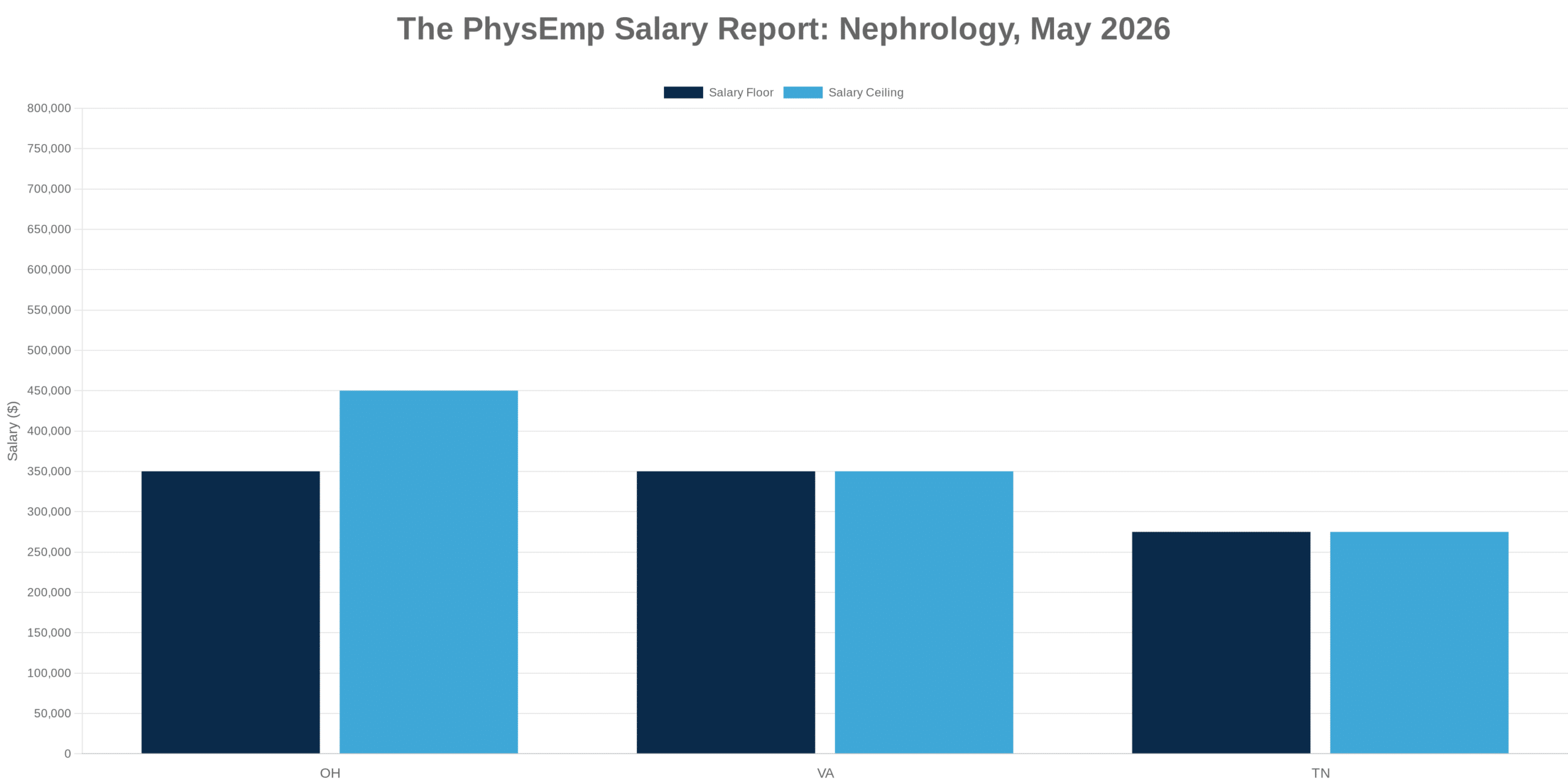
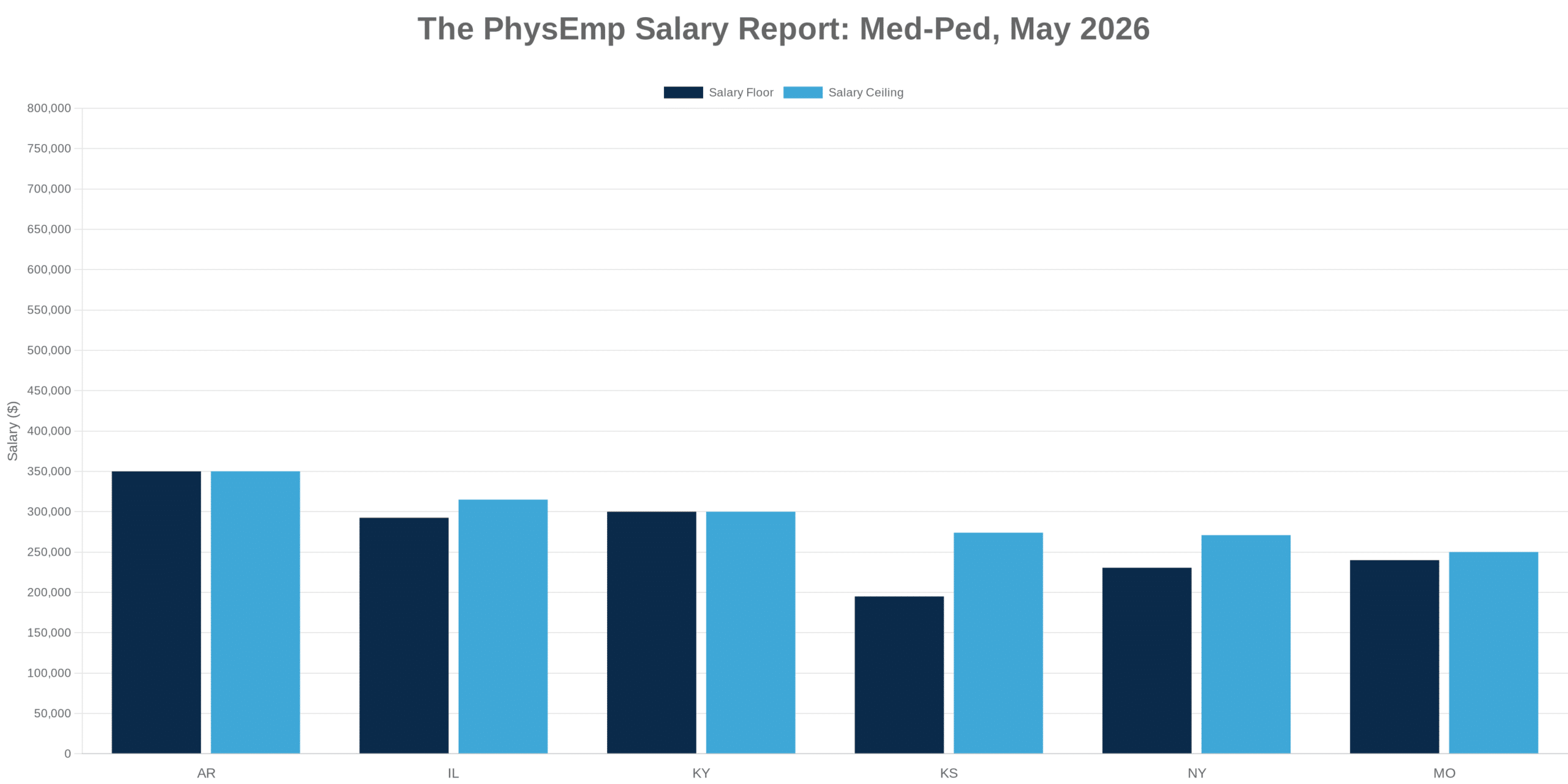
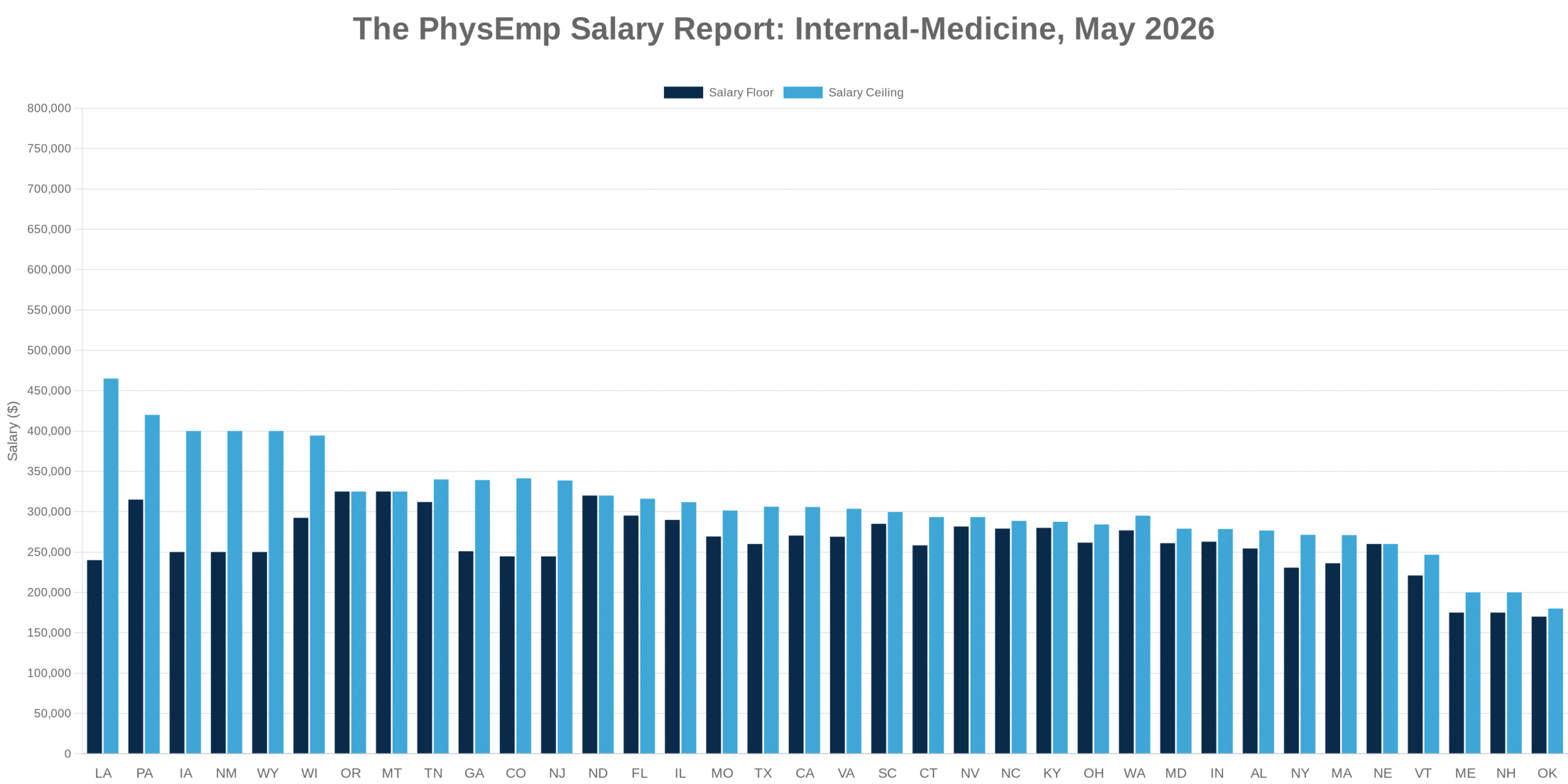
Why This Theme Matters Now
The integration of artificial intelligence (AI) in the pharmaceutical sector has reached a critical juncture where the demand for faster drug development coincides with evolving regulatory landscapes. In an era where timely access to medications can influence health outcomes, the adoption of AI technologies is not only transforming traditional methodologies but is also reshaping the very frameworks of clinical trials and regulatory submissions—marking a broader shift in AI in healthcare. This shift is particularly relevant as we approach 2026, a year designated by industry experts as a turning point for clinical operations.
AI Enhancements in Clinical Trials
According to a report by Reuters, drugmakers are increasingly relying on AI to expedite clinical trials. These technologies enable companies to analyze vast amounts of trial data more efficiently and accurately, allowing for the identification of optimal patient populations and refining trial designs. By doing so, pharmaceutical firms can significantly shorten the timelines traditionally associated with drug development. The prospect of AI not only streamlining these processes but also enhancing data quality raises crucial questions about its impact on scientific integrity and patient safety.
Rethinking Regulatory Submissions
The implications of AI in regulatory submissions are profound. AI tools facilitate the rapid preparation of regulatory documents, thus potentially accelerating the approval processes by providing regulators with clearer and more relevant data. However, as highlighted by Applied Clinical Trials, this rapid advancement is accompanied by the necessity for regulatory bodies to develop stringent evaluation frameworks for AI-assisted submissions. Because AI is a continually evolving domain, establishing robust standards will be essential for ensuring that the medicines emerging from these processes are safe and effective.
Best Practices for AI Implementation
In the quest for effectively integrating AI into clinical operations, organizations face the challenge of maintaining scientific rigor while pursuing innovation. Best practices emerging from recent discussions in the field emphasize the importance of fostering collaboration between data scientists and clinical professionals. This synergy can drive the development of tools that not only enhance patient recruitment processes but also ensure compliance with safety standards. Supporting this endeavor requires an investment in training and infrastructure as organizations navigate the complex landscape of AI technologies.
Balancing Innovation and Compliance
As pharmaceutical companies embrace AI, a careful balance must be struck between advancing technology and adhering to regulatory requirements. The responsibility lies not only with drug manufacturers but also with regulatory agencies. Ensuring that AI solutions do not compromise the quality or safety of clinical trials remains paramount as the industry moves forward. It is crucial for all stakeholders to engage in an ongoing dialogue aimed at refining both the technological capabilities and the regulatory frameworks that govern them.
The use of AI in clinical trials represents a paradigm shift that promises to enhance efficiency, yet it necessitates a holistic approach to compliance and patient safety, ensuring that innovation does not come at the expense of regulatory integrity.
Implications for the Healthcare Industry
The transformation brought about by AI in clinical trials and regulatory submissions has significant implications for the healthcare industry, particularly in the context of recruitment and talent acquisition. As the demand for talent capable of bridging clinical and technological domains increases, so too does the importance of creating pathways for continuous professional development in this field.
Conclusion
The advances in AI technologies present unprecedented opportunities to enhance the efficiency and effectiveness of clinical trials and regulatory processes. However, as we look to 2026 and beyond, it is vital for the pharmaceutical industry to prioritize the development of thoughtful regulatory standards that ensure patient safety is upheld. Balancing innovation with compliance will be critical in shaping the future of healthcare.
Sources
Drugmakers turn to AI to speed trials, regulatory submissions – Reuters
What 2026 Will Demand From Clinical Operations: Rethinking Efficiency, Access, and AI – Applied Clinical Trials