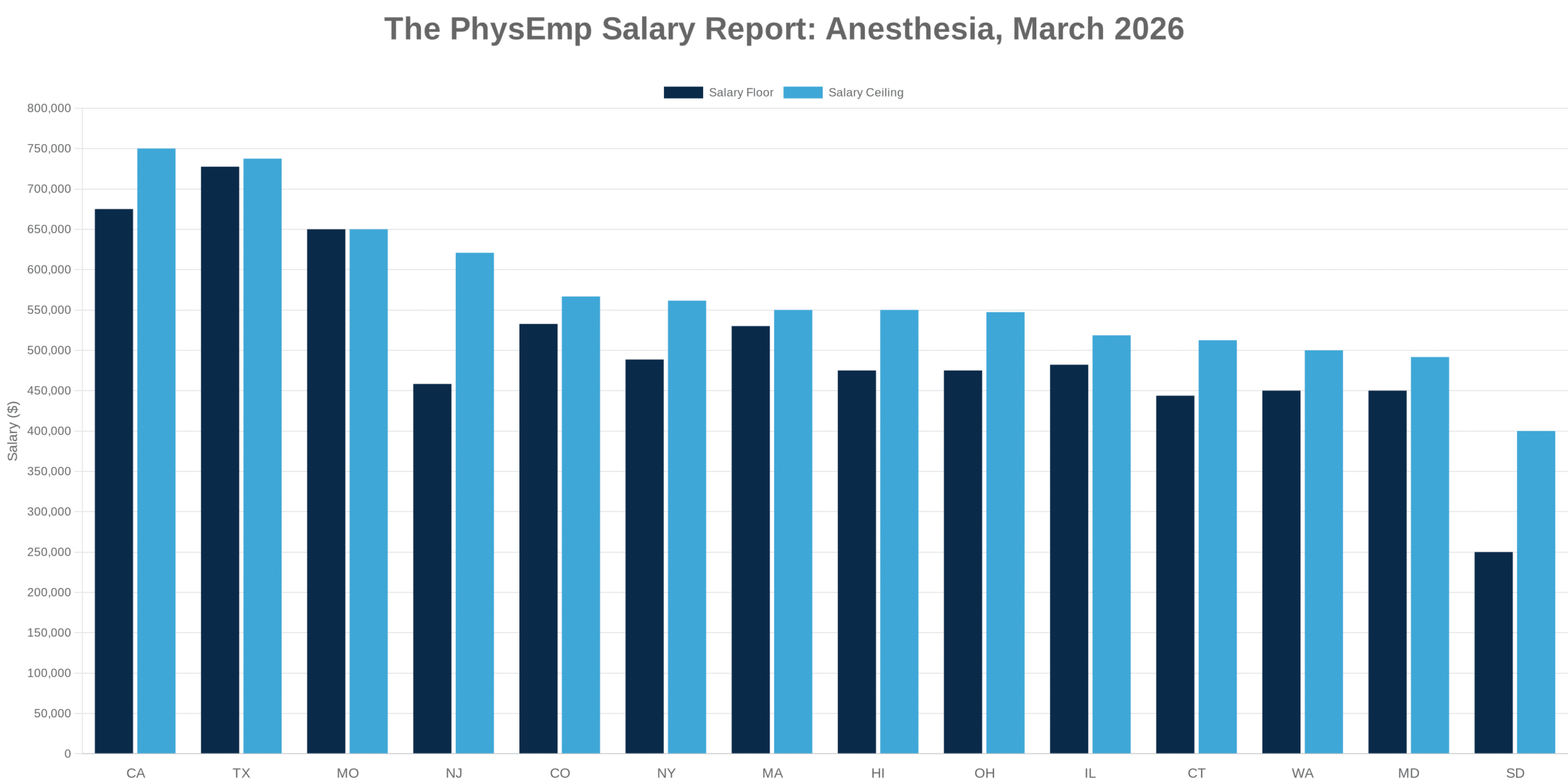
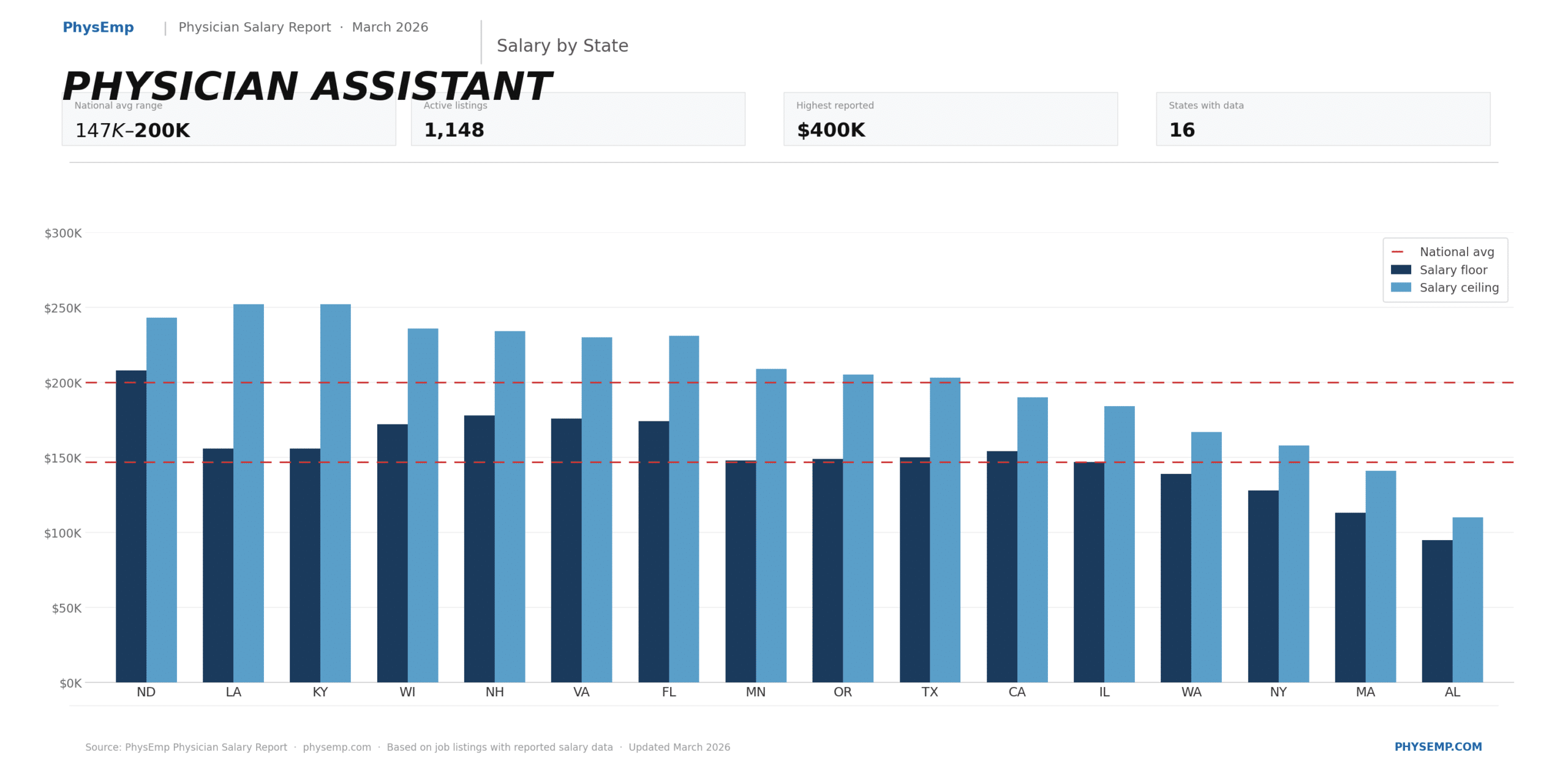
Why this theme matters now
Healthcare organizations and federal agencies are deploying artificial intelligence faster than structured Ai inAI in healthcare can evolve to oversee it. That acceleration—combined with reports of automated decision-making in prior authorization, growing internal AI use within regulators, and calls for stronger board-level oversight—creates a convergence of operational, safety, and reputational risk. When algorithmic choices are neither explainable nor auditable, patients face delayed or denied care, clinicians shoulder unaccountable workflow burdens, and institutions inherit legal and financial exposure. The current moment demands attention because adoption is outpacing transparency: systems that shape access to care are already in production even as accountability mechanisms lag.
Opacity in prior authorization: operational and patient risks
Prior authorization workflows are traditionally opaque and resource-intensive; introducing AI can scale those properties rather than fix them if models are not transparent. Automated rules or learned models that screen authorization requests may amplify biases embedded in training data, produce inconsistent denials across similar clinical scenarios, and put clinicians into protracted appeals processes. From a systems perspective, opaque algorithms translate to unpredictable capacity demands—more administrative load, longer patient wait times, and inconsistent outcomes for beneficiaries. Where Medicare Advantage plans or intermediaries rely on non-interpretable models, the consequences are not just efficiency losses but clinically meaningful delays in care.
Regulatory agencies increasing AI use while disclosure lags
Public-sector adoption of AI for oversight, internal triage, and program operations is expanding. Yet many agencies have not codified requirements for documentation, public reporting, or external audit of the tools they use. This asymmetric disclosure—agencies using AI but failing to publish model purposes, risk assessments, or performance metrics—creates a governance blind spot. Without baseline transparency, policymakers and affected stakeholders cannot assess whether an algorithmic intervention improves outcomes, preserves equity, or introduces new failure modes. The practical implication is that public trust in regulatory decisions may erode even when intent is improvement, because the mechanisms producing outcomes remain hidden.
Call Out: Transparent design and traceable decision paths are not optional safety features; they are preconditions for durable public trust. Health systems that treat explainability and auditability as core design constraints reduce downstream legal, operational, and patient-safety costs.
Board-level accountability and vendor transparency
Corporate governance influences how quickly transparency advances. Boards that accept opaque vendor claims or defer oversight to procurement teams miss an opportunity to shape safer deployment. When major technology providers resist substantive disclosure—citing intellectual property or competitive advantage—boards must weigh those claims against fiduciary exposure from adverse events, privacy breaches, or regulatory sanctions. Effective governance requires contractual levers: mandatory model documentation, rights to third-party audits, incident reporting clauses, and clear acceptance criteria for clinical deployments. Without those levers actively exercised at the board level, organizational exposure concentrates in operations and clinical risk managers.
What governance practices materially reduce risk
– Standardized documentation (model cards, data provenance, versioning)
– Independent technical and clinical audits before wide deployment
– Human-in-loop thresholds for high-stakes decisions
– Performance monitoring with real-world metrics and bias checks
– Contract clauses granting audit rights and incident notification timelines
Reconciling innovation with accountability: operational paths forward
Moving from principle to practice means aligning procurement, clinical leadership, and compliance on minimum transparency standards. Organizations should require vendors to provide model documentation, validation datasets or summary statistics, and an auditable decision trail. Internally, multidisciplinary review committees—comprising clinicians, data scientists, ethicists, and compliance officers—should sign off on risk assessments and acceptable failure modes. Regulators can accelerate alignment by requiring public disclosure of agency AI use and by establishing baseline reporting standards for health outcomes and fairness metrics.
Call Out: Hiring for governance competence—roles such as ML auditors, clinical AI risk managers, and regulatory liaisons—reduces implementation risk and signals to stakeholders that the organization treats AI accountability as a strategic priority.
Implications for the healthcare industry and recruiting
The accountability gap creates demand for a new set of competencies across healthcare employers. Recruiters and hiring managers should anticipate increased need for professionals who can translate clinical risk into technical specifications: model risk officers, explainability engineers, clinical safety reviewers for AI, and compliance leads with ML literacy. These roles bridge the divide between innovation and governance and will be critical to restoring stakeholder trust.
Conclusion: recalibrating trust through accountable design
AI can improve throughput, detect patterns, and support clinical decisions—but those benefits are conditional on transparent, auditable design and active governance. The current mix of opaque prior authorization systems, under-disclosed agency AI use, and lagging board accountability creates systemic risk. Addressing that risk requires operational standards, contractual transparency, independent evaluation, and a workforce equipped to manage AI-specific governance tasks. The longer transparency gaps persist, the greater the erosion of trust—and the harder it will be to realize AI’s potential in healthcare without regulatory backlash or patient harm.
Sources
US health agency ramps up AI use as transparency gaps persist – The Pharma Letter
With AI accountability stalling, boards must push tech giants for greater transparency – Reuters