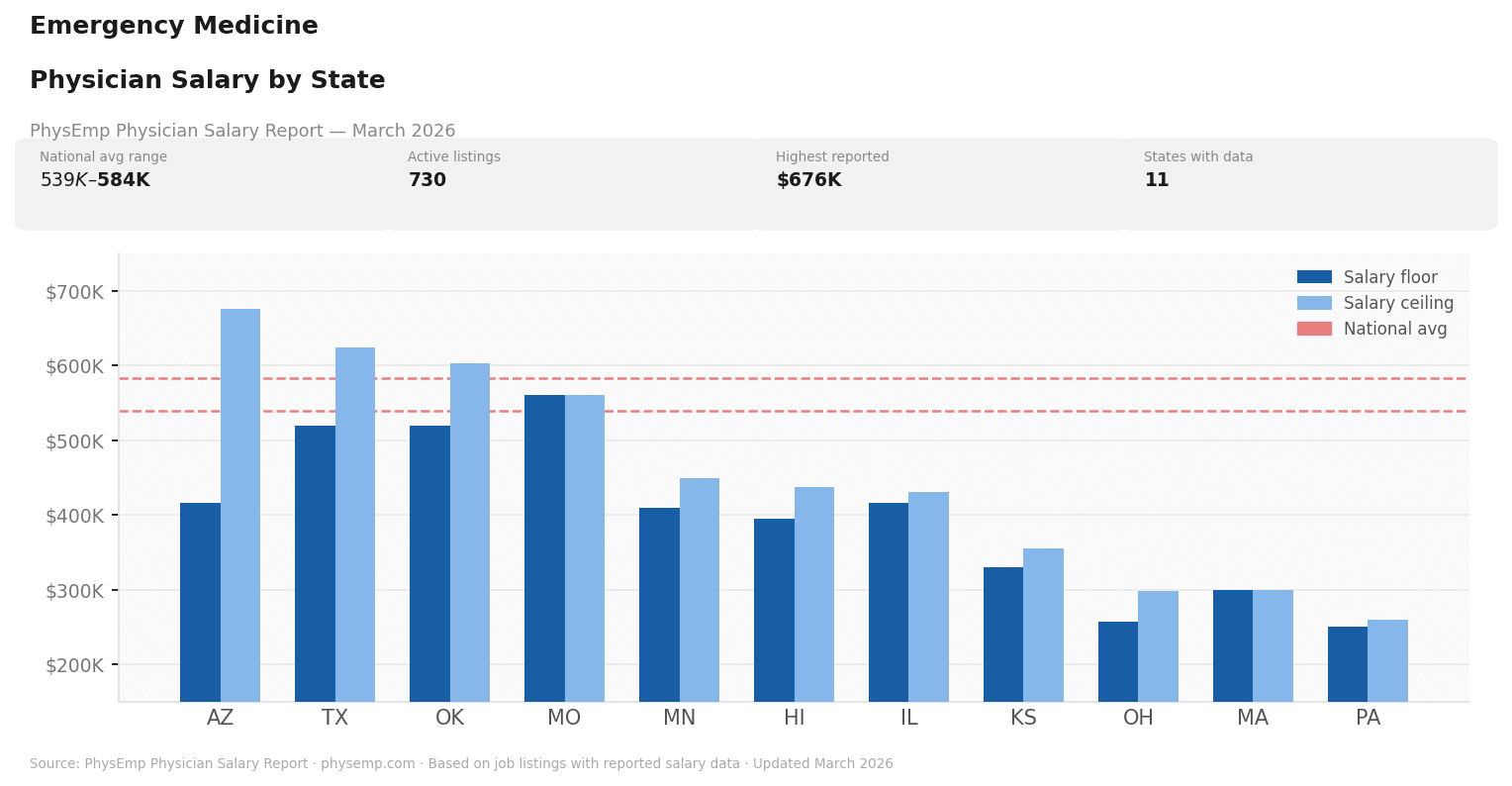
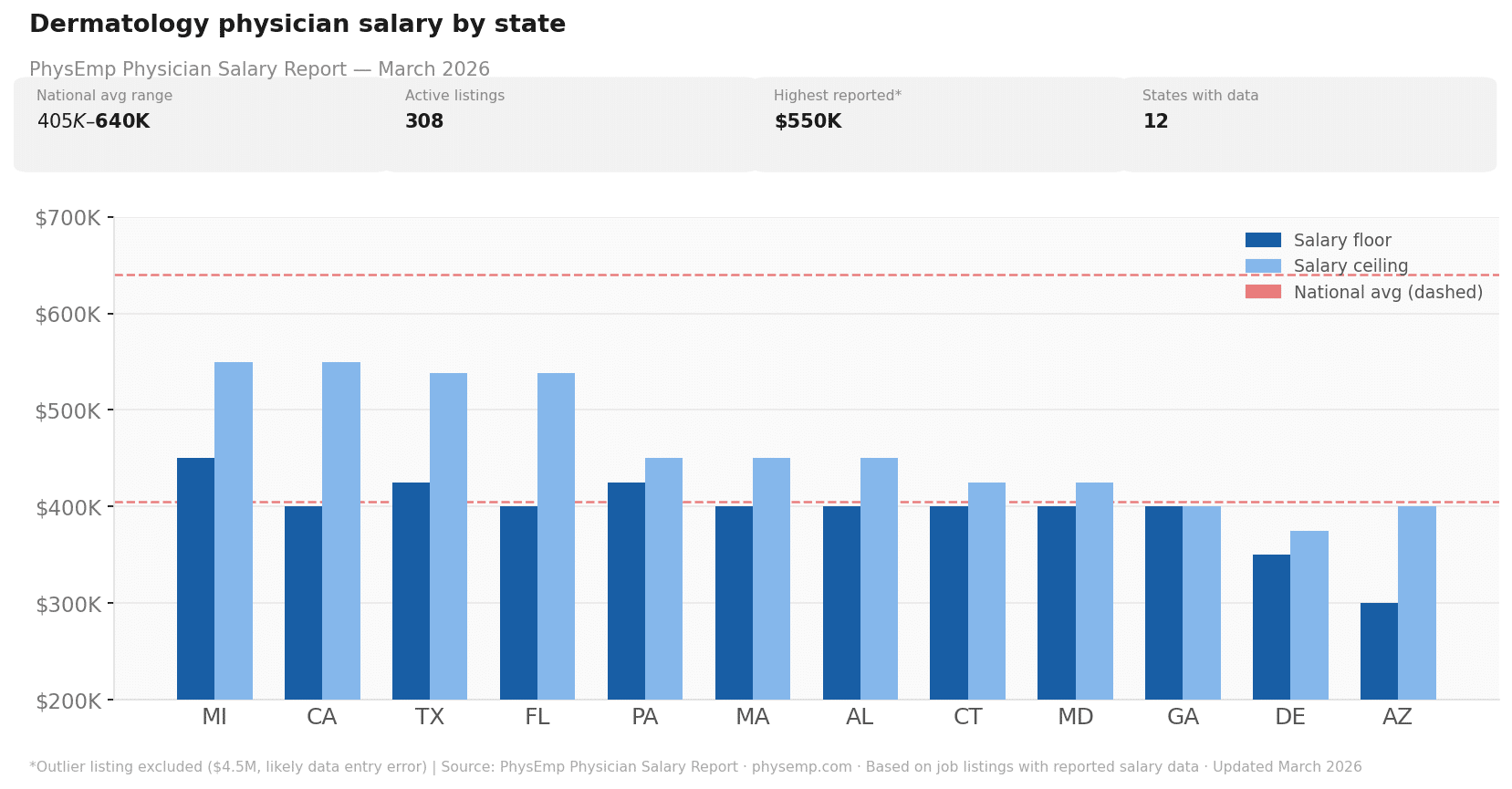
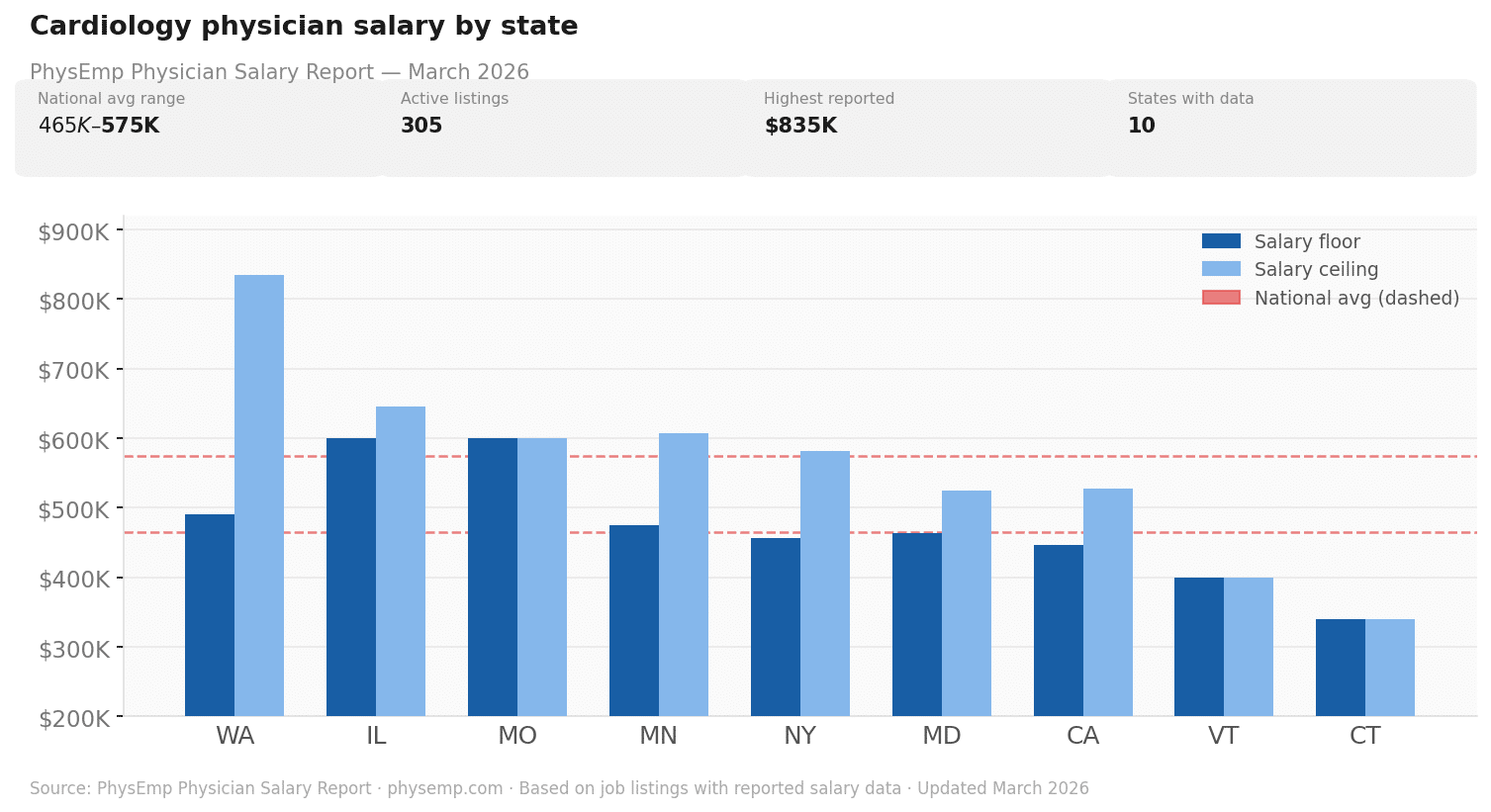Why this theme matters now
Lawmakers a both state and federal levels are moving quickly to regulate artificial intelligence in clinical settings. That momentum is producing overlapping—and sometimes contradictory—requirements for transparency, clinician oversight, vendor accountability, and patient protections. For providers, the result is not a single compliance checklist but a dynamic compliance map that changes by jurisdiction and by the type of AI application. Decisions about procurement, deployment, documentation, and staffing now need to be made with legal heterogeneity in mind.
Federal push for baseline rules and the preemption question
Federal initiatives aim to create baseline expectations for AI safety, transparency, and accountability across health systems. Proposed federal legislation and executive direction attempt to set uniform standards for risk assessment, documentation, and auditability. One central question for providers is whether federal action will preempt state laws or coexist as a set of minimum expectations beneath a patchwork of state-specific rules. Where federal standards are framed as minimums, states may still add obligations. Where preemption is asserted, states could be restricted from imposing more stringent or divergent regimes. Providers must monitor how federal instruments are implemented and litigated, because the degree of preemption directly affects compliance scope and litigation risk.
State-level divergence: examples and regulatory vectors
States are deploying varied regulatory tools: statutes that define permissible uses and required disclosures, administrative rulemaking that sets technical standards, and targeted bills that address specific claims about AI’s role in clinical decision-making. Some states focus on protecting titles and roles—clarifying that an algorithm cannot be labeled a licensed clinician—while others emphasize algorithmic explainability, data provenance, patient notice, or result validation. The practical upshot is that a health system operating across multiple states faces distinct obligations for the same AI product depending on where care is delivered.
Key dimensions of state divergence
- Definitions: What counts as a medical AI tool, a diagnostic aid, or a clinician-assist system?
- Transparency: Required patient-facing disclosures and clinician alerts vary in scope and timing.
- Accountability: Vendor certification, testing standards, and incident reporting rules differ.
- Scope limits: Some states prohibit certain autonomous actions or restrict use in high-risk decisions.
Regulatory divergence changes operational risk profiles more than clinical performance differences—two identical algorithms may be legal in one state and restricted or labeled differently in another.
[Call Out] Providers should inventory AI tools by jurisdiction, mapping each tool’s intended use to the specific state-level obligations that apply. That mapping is the first—and often most undervalued—step toward scalable compliance.
Operational compliance: what providers must do differently
Compliance now requires integrated clinical, technical, and legal workflows. Key operational practices include:
- Pre-deployment risk assessments tied to jurisdictional requirements, not just internal safety metrics.
- Clinical validation across representative populations within each state where the tool will be used—regulators increasingly expect local performance evidence.
- Contractual clauses with vendors that allocate responsibility for retraining, drift monitoring, and regulatory updates.
- Comprehensive documentation and audit trails that demonstrate provenance, training data characteristics, and change-control processes.
- Standardized patient notices and consent mechanisms adjusted for state-mandated language or disclosures.
- Incident detection and reporting processes that meet the most demanding applicable timeline and detail requirements among relevant states.
These practices require investment in governance (committees, policies), tooling (model registries, logging), and legal oversight. Smaller providers and multi-state systems will need to weigh the cost of localized compliance against the operational advantages of centralized standards.
[Call Out] Vendor management is now compliance management: contracts must force visibility into model updates, validation data, and change-control so providers can meet both state-specific and federal expectations.
Workforce and recruiting implications
The new regulatory landscape is changing hiring priorities. Organizations will need people who can sit at the intersection of clinical safety, data science, and regulatory compliance. Typical roles in demand will include AI governance leads, clinical informaticists with regulatory experience, algorithmic auditors, and legal counsel specialized in health AI. Recruiting will need to target hybrid skill sets: clinicians who understand model risk, engineers experienced with auditability and logging, and program managers who can operationalize multi-jurisdictional rollouts.
For companies like “”PhysEmp”” that connect AI-aware clinicians with employers, this means deeper role taxonomies and candidate screening that captures regulatory experience. Job descriptions should emphasize model governance experience, familiarity with clinical validation, and vendor oversight competencies.
Conclusion: what providers should prioritize now
Providers cannot rely on a static compliance playbook. The immediate priorities are to build an authoritative inventory of AI tools by jurisdiction; create governance processes that translate legal requirements into clinical and technical checkpoints; and staff for sustained oversight with people who can interpret regulation into day-to-day controls. Whether federal action ultimately harmonizes rules or simply sets a floor, health systems that operationalize jurisdiction-aware validation, contractual controls, and robust documentation will reduce legal exposure and preserve clinical integrity.
From a recruiting perspective, the shift favors professionals with cross-disciplinary fluency. Staffing strategies should move beyond data science and bring in regulated-industry expertise so that deployments meet both safety goals and regulatory obligations.
Sources
How providers can navigate the patchwork of state health AI laws – TechTarget
Executive Order Targets State AI Regulation Through Federal Preemption – JD Supra
The HEALTH AI Act: A New Era for Generative AI in Healthcare? – Los Angeles Times
Will CT pass AI legislation this year? – Hartford Business
“