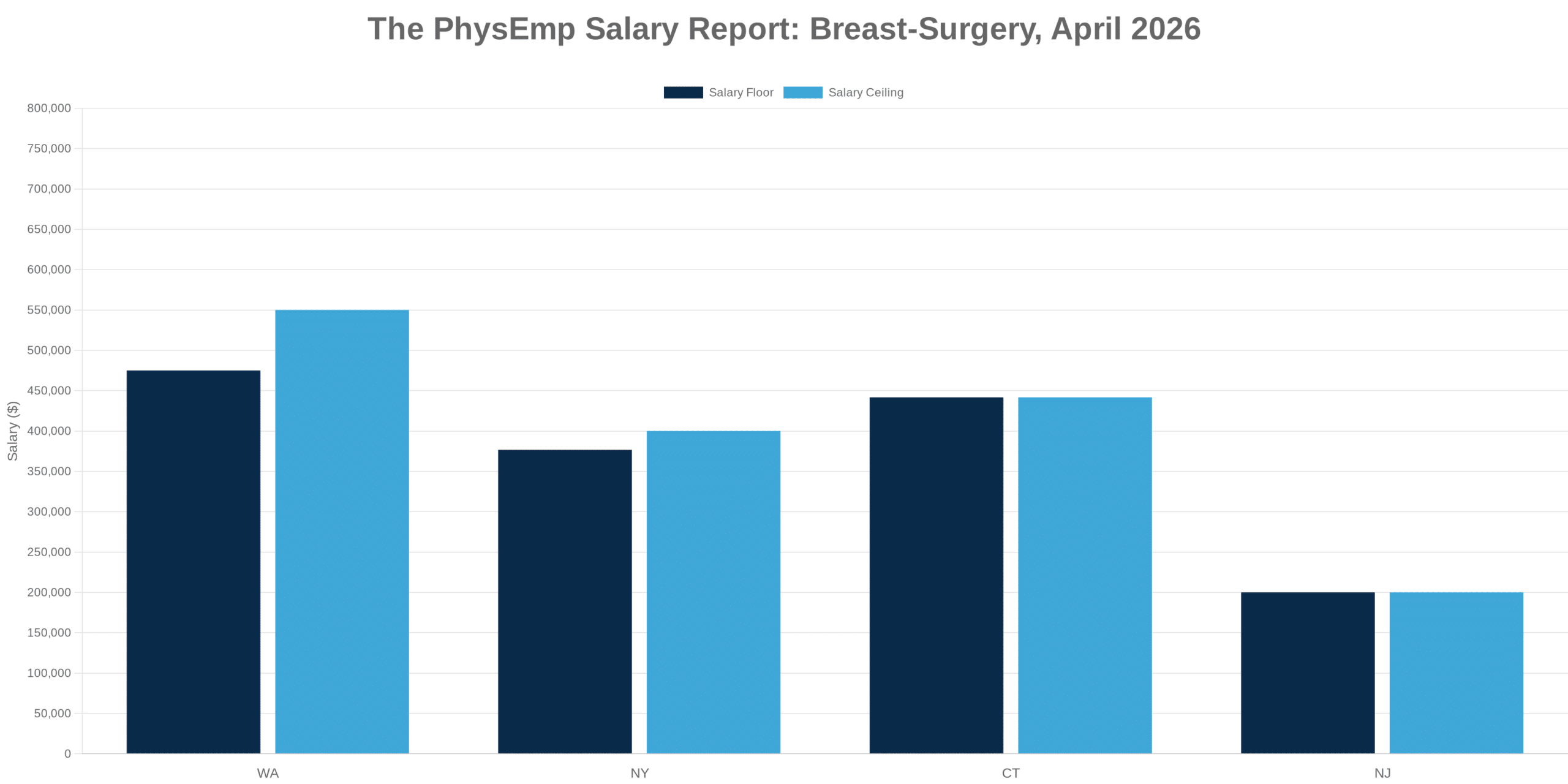
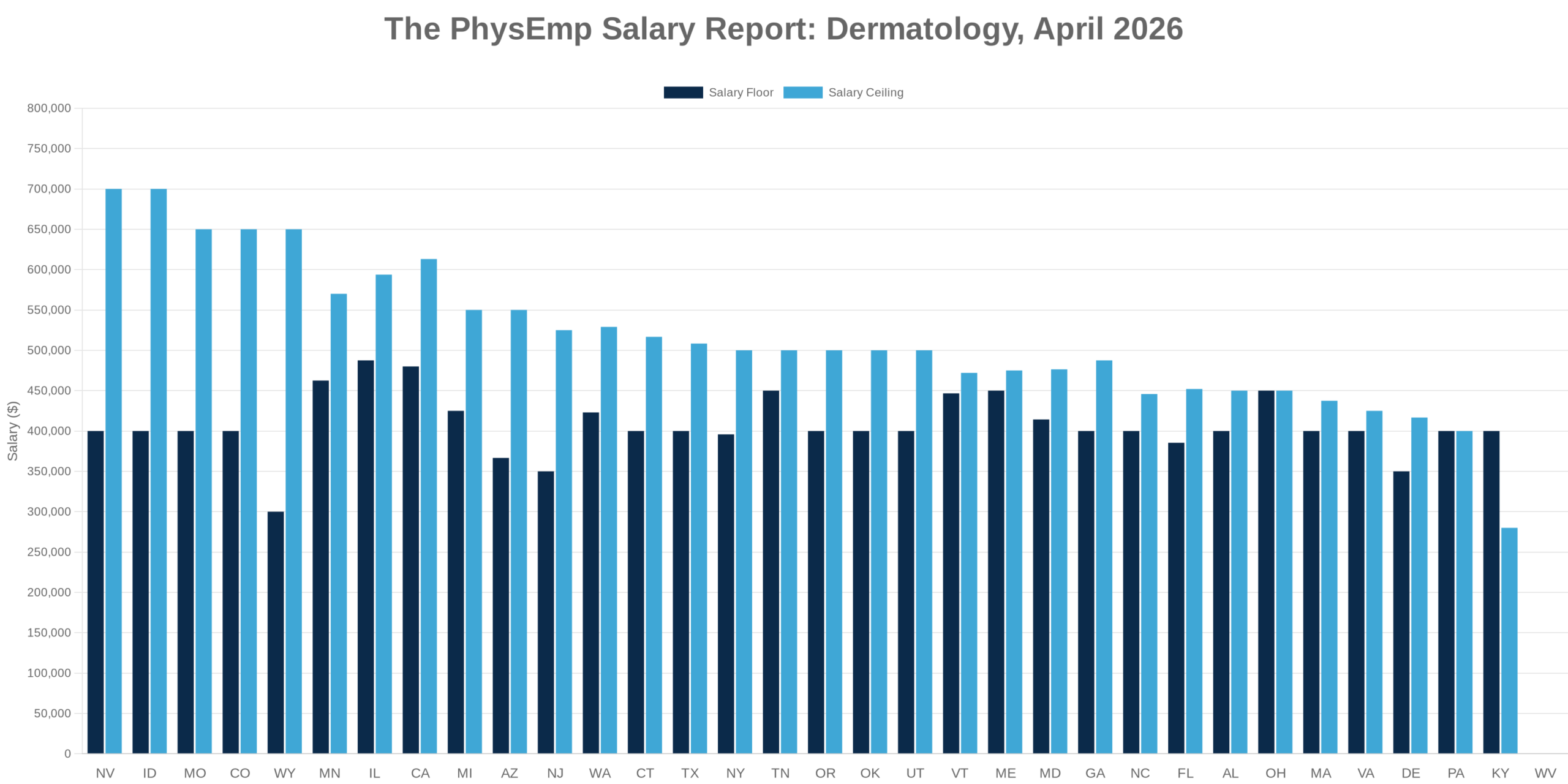
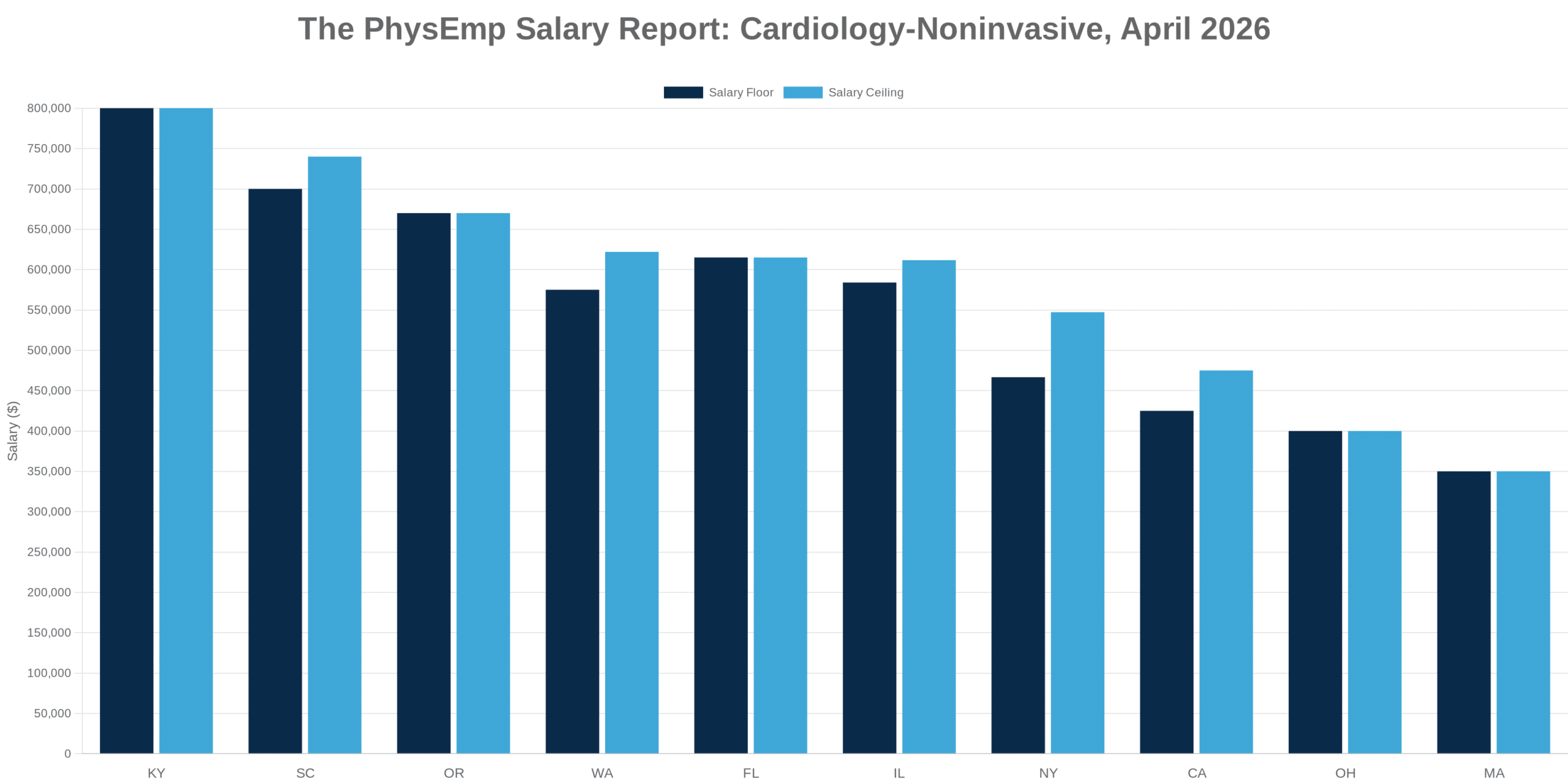
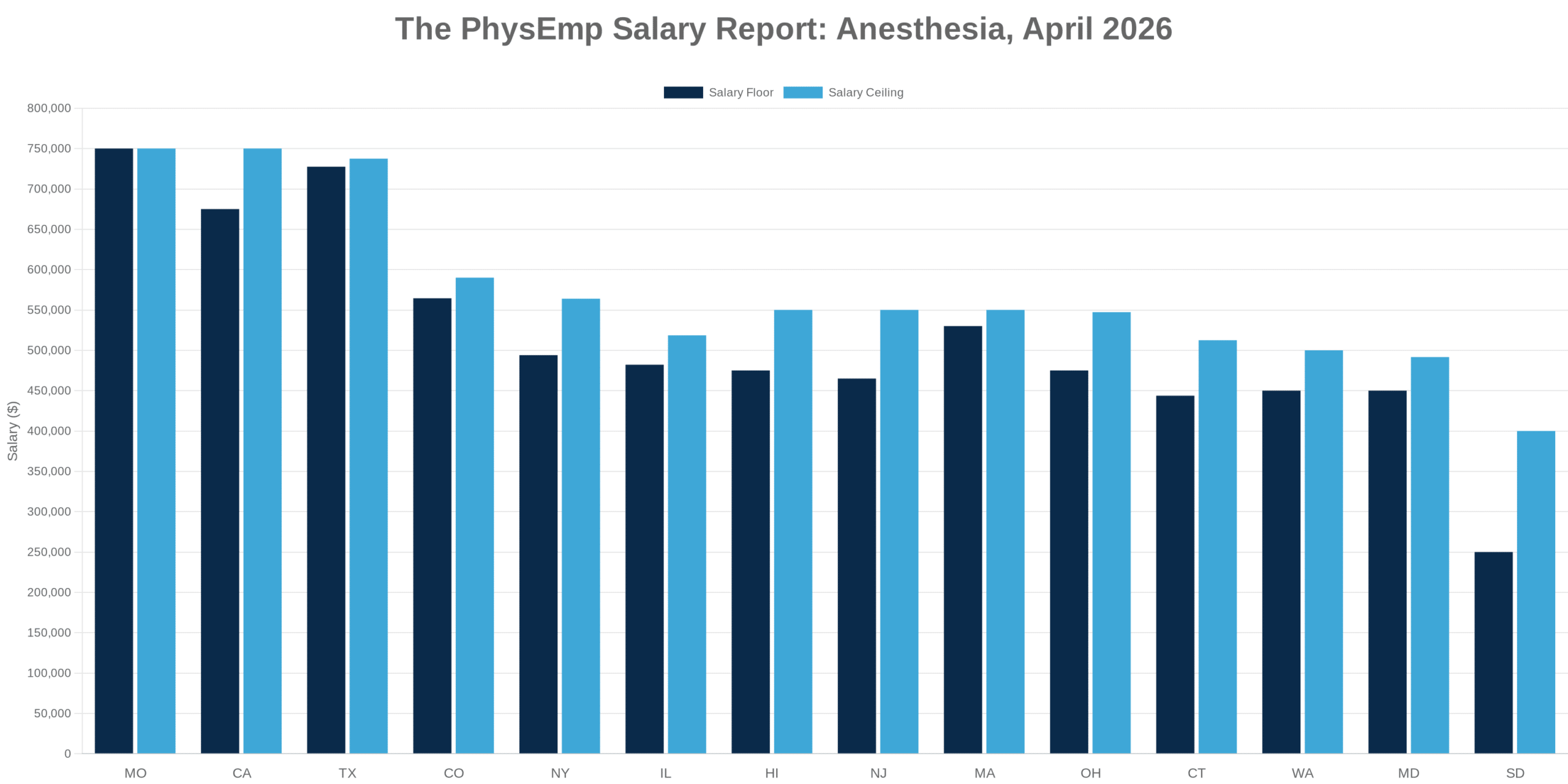Why this theme matters now
Artificial intelligence firms are expanding their influence beyond product development and commercialization into the domain of public policy funding. A recent multimillion-dollar contribution from a leading AI developer to a policy research organization reflects a broader pattern: private capital is increasingly supporting the research that informs how regulators, payers, and health systems oversee algorithmic tools.
In healthcare—where patient safety, equity, and clinical accountability are directly implicated—the governance of AI may be shaped not only by technical validation but also by who finances the policy discourse. The growing intersection of industry funding and regulatory design raises material questions about independence, transparency, and the balance between innovation advocacy and patient protection.
These dynamics sit squarely within the broader transformation of AI in Physician Employment & Clinical Practice, where governance frameworks determine how AI-enabled care is integrated into clinical workflows, employment structures, and long-term workforce strategy.
Who is stepping in — and why it matters
Large AI vendors face immediate pressure to reassure stakeholders that their systems are safe, fair, and auditable. One way they respond is by channeling resources into policy research organizations and think tanks. The stated intent is to support evidence development, stakeholder outreach, and regulatory literacy. For health systems and regulators, this influx of funding can accelerate needed studies on clinical risk assessment, model validation standards, and workforce impacts. But because those studies can also inform the rules under which vendors operate, the source of funding can shape research questions, methods, and dissemination priorities.
Call Out — Independence Risk: When private actors underwrite policy work, the potential for agenda-setting increases: funders may prioritize pragmatic regulatory outcomes that favor rapid deployment over conservative safety checks. Healthcare stakeholders must insist on transparency about funding streams and governance of funded projects.
Influence pathways: how funding can shape governance
There are several mechanisms through which vendor funding can affect policy outcomes. First, financial support can determine which topics receive study — prioritizing usability and market adoption issues over long-term safety or equity evaluations. Second, funded research programs often organize convenings and advisory groups, which can shape the composition of voices at the table. Third, sustained funding relationships create reputational ties between researchers and benefactors, subtly influencing framing, interpretation, and publication choices.
In healthcare, these influence pathways matter because regulatory frameworks are still emerging. Rules on clinical validation, data provenance, and liability will be contested — and research that informs those debates must be defensible. If the evidence base is perceived as industry-influenced, payers, clinicians, and patient advocates may distrust recommended pathways, slowing adoption or prompting more prescriptive regulation.
Opportunities: capacity building and evidence gaps
Private funding is not inherently problematic. Health policy research has long relied on mixed funding models, and new resources can fill critical capacity gaps: larger sample studies, algorithmic audit programs, cross-institutional datasets, and accelerated translation of technical findings into usable policy recommendations. For healthcare, these investments can enable pragmatic work on interoperability standards, model monitoring protocols, and clinician workflow integration.
Call Out — Strategic Opportunity: Directed, transparent funding can accelerate development of practical governance tools — for example, standardized clinical performance metrics and post-market monitoring approaches — reducing friction between innovation and patient safety.
What this signals for AI governance in healthcare
The participation of AI companies in funding policy research signals a maturing industry that recognizes governance as a competitive and reputational concern. It also reflects a strategic shift: vendors are investing in the institutional architecture that will determine market access. For regulators and health systems, this duality requires careful management. Engagement with vendor-funded research should be conditional on clear safeguards: public registration of funded projects, pre-specified research protocols, independent peer review, and publication commitments regardless of outcome.
Implications for healthcare organizations and recruiting
For healthcare providers, payers, and clinical research groups, the development has concrete hiring and capability implications. Organizations will need staff who can translate policy research into operational governance: clinical AI safety officers, regulatory program managers, data governance leads, and legal professionals versed in algorithmic liability. Recruiters will see rising demand for hybrid profiles that combine clinical domain knowledge, AI literacy, and policy experience. The market will favor candidates comfortable navigating technical validation, stakeholder convening, and compliance with evolving regulatory standards.
For AI job boards and talent platforms, there is an opportunity to surface roles tied to governance and policy. Candidates who can bridge technical evaluation and policy translation will be scarce and in high demand. Platforms that curate these roles and highlight track records in impartial, interdisciplinary research will provide strong value to both employers and applicants.
Practices to preserve research integrity
To ensure that vendor-funded policy research enhances governance rather than distorts it, stakeholders should adopt a set of best practices: mandatory disclosure of funding sources; independent advisory or steering committees with public-interest representation; pre-registration of research questions and methods; commitments to open data or reproducible workflows where privacy allows; and clear conflict-of-interest management for authors and conveners. These safeguards help maintain credibility, enable critical appraisal, and ensure policy debates are informed by verifiable evidence.
Conclusion — balancing influence and impact
Industry funding of public-interest policy research is neither neatly virtuous nor inherently suspect. For healthcare, the stakes are too high for indifference: funding can accelerate urgently needed governance capacity, but without rigorous transparency and safeguards it risks shaping policy in ways that prioritize market access over patient protection. Policymakers, funders, health systems, and hiring managers must therefore treat these investments as strategic collaborations requiring clear guardrails. The emerging workforce must include professionals adept at managing those guardrails — a demand that will reshape recruiting priorities across healthcare and medical AI.